Shape Up: a complete guide to this new development methodology (2023)
The Shape Up method enables engineering teams to optimize their workflow, improve output, and bring sanity into the development process.

Shape Up is a project management methodology developed by Basecamp. Shape Up brings time-boxed iterative delivery with lightweight upfront shaping. The goal is to build the right things through rapid experimentation and learning rather than big, upfront planning.
Welcome to your complete guide on adopting Shape Up, a revolutionary new approach to project management.
Shape Up helps teams build better products in less time.
In the next 15 minutes I'll break down everything you need to know.
Ready to be an expert?
Let's get started.
If you've been using Agile frameworks like Scrum or Kanban and feel they're no longer working for your team, Shape Up may be the solution you've been searching for.
The Shape Up Method is instrumental in creating many successful products, including the iPhone, Tesla Model 3, Amazon Kindle, and Basecamp.
I'll walk you through what makes Shape Up different...
- The core principles and concepts
- The practice and stages that make it so effective, and with...
- Step-by-step instructions for you to implement it successfully
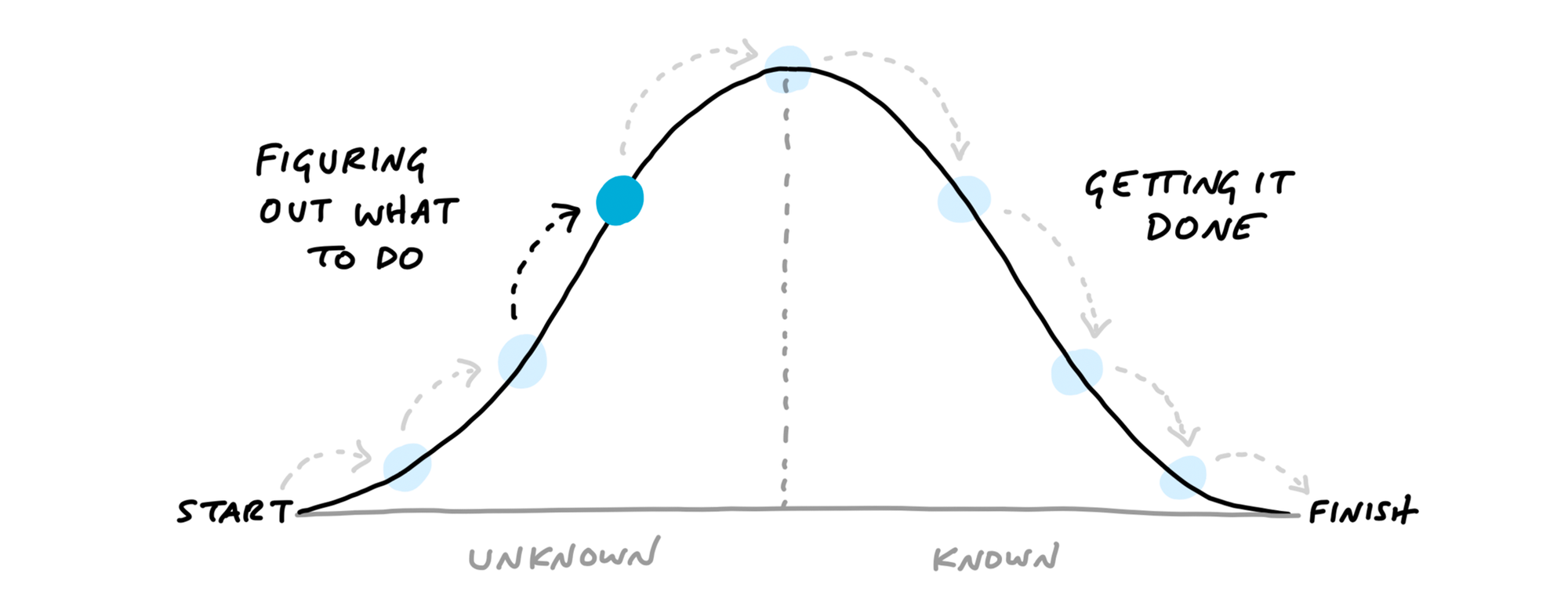
You'll transition to 6-week cycles focused on solving meaningful problems, quickly map out solutions instead of getting bogged down in specification, and limit Work in Progress to enable greater productivity and focus.
Shape Up offers a refreshing new take on product development that replaces prescriptive processes with simple principles for making smart bets.
It enables teams to adapt quickly, focus efforts, and deliver meaningful results frequently.
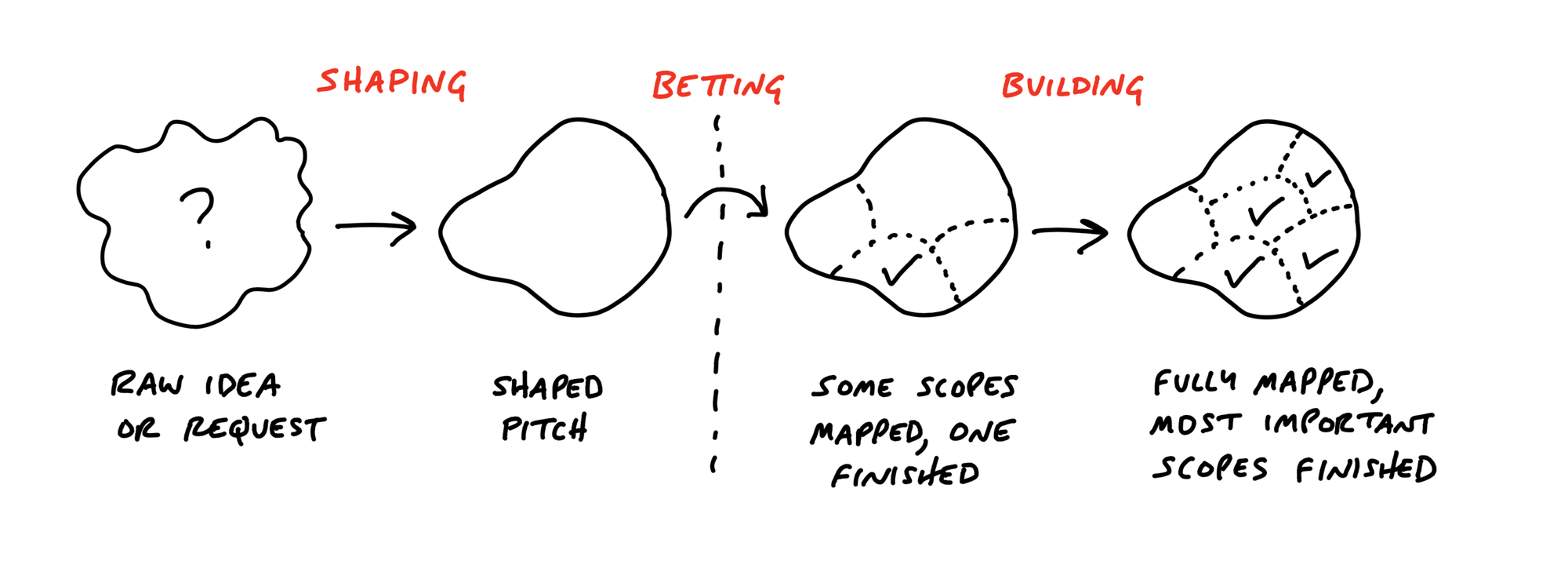
Key Takeaways
If you stop reading in the next minute, and aren't ready to commit another 10 to complete the guide, here's what you need to know about Shape Up...
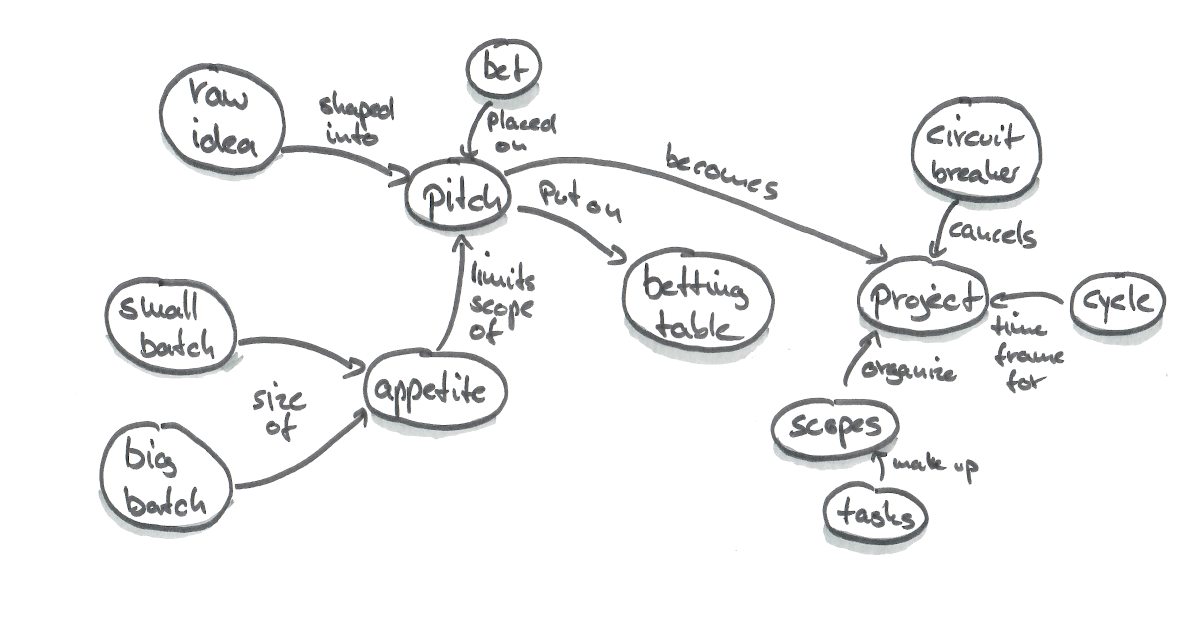
Cycles
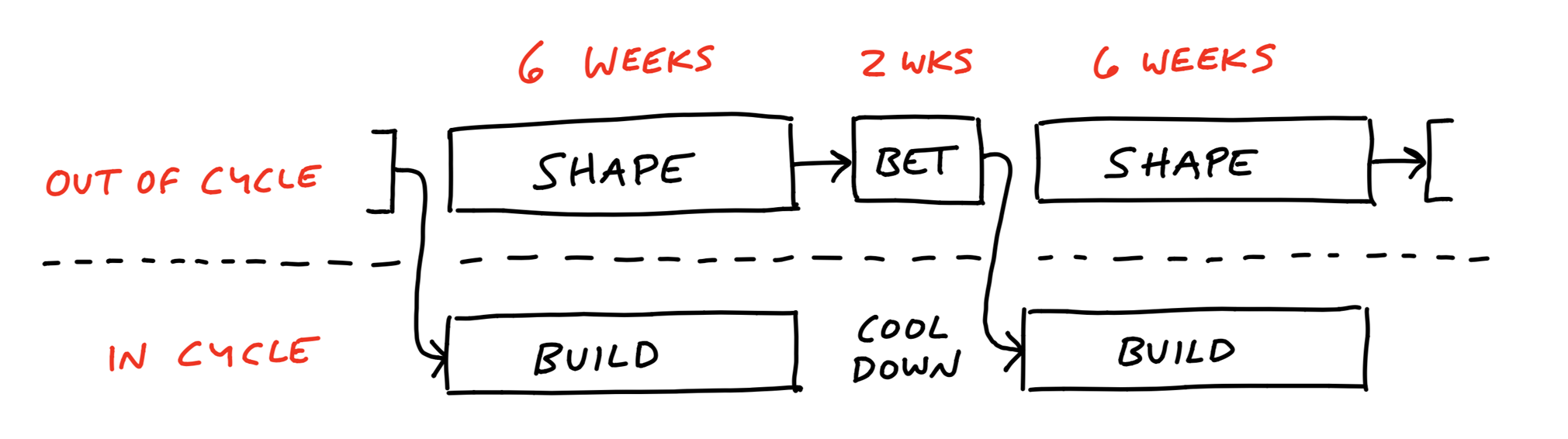
Six-week cycles provide fixed timeboxes for teams to complete meaningful work rather than open-ended projects. This timeline forces focus and helps prevent scope creep.
Shaping
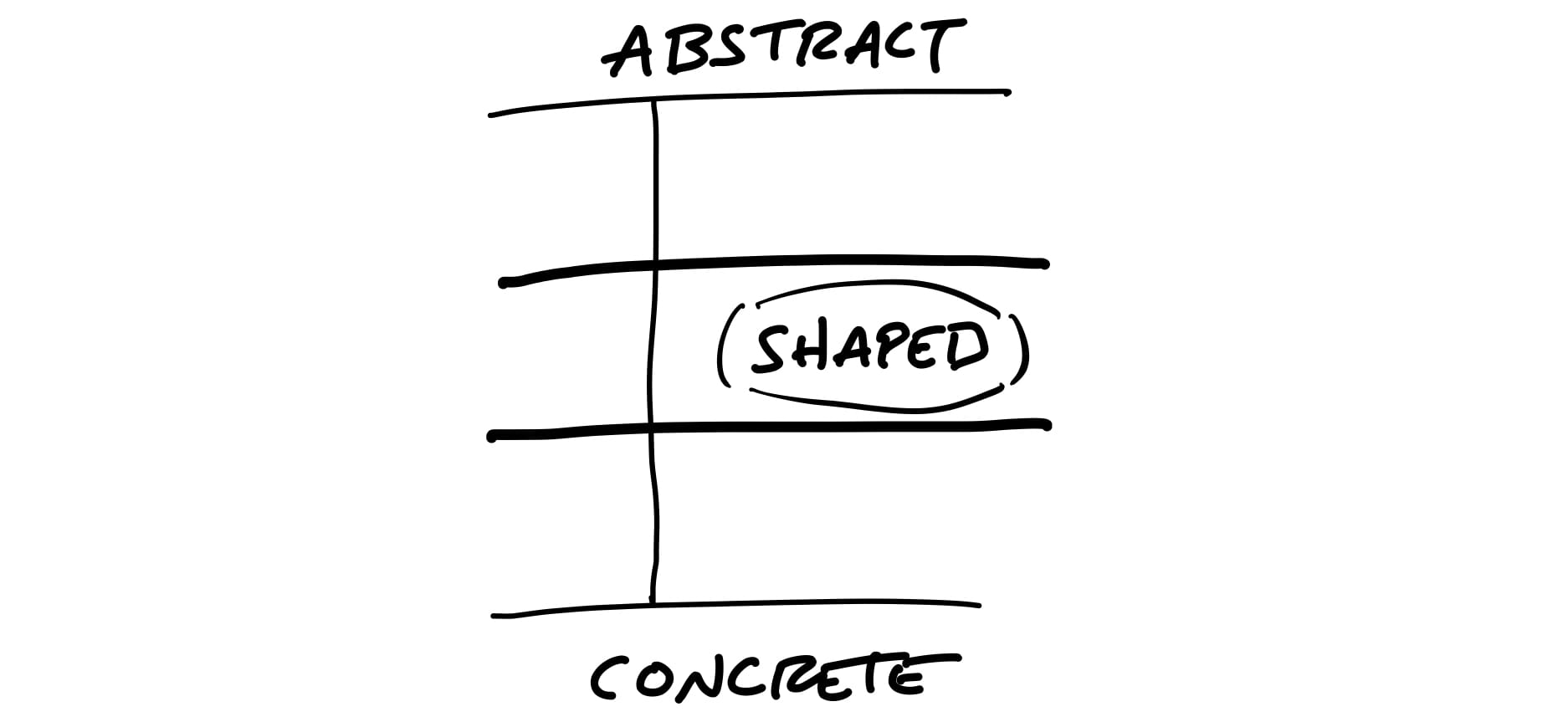
Shaping upfront resolves ambiguities early so teams don't waste time clarifying details later on. Shaping involves high-level framing of the problem and desired outcomes.
Betting

Betting is how leadership aligns on priority projects for each cycle. Betting focuses efforts on the initiatives that matter most to advance strategic goals. Thinking bets moves you to allocating time as money, rather than scoping infinite projects.
Ready to get shaping?
If you're ready to level up your product development practices, then let's get started on how Shape Up can transform the way your team works.
With the strategies in this guide, you'll be set up for success no matter what challenges your projects throw your way.
Now – let's get you shaping...
The founding team behind this blog uses Shape Up to build Klu. Need help with your AI Strategy? Grab time with Stephen, he'll be happy to help.

Shape Up: How Does It Work?
Shape Up is a product development company that uses a unique approach to help businesses improve their products. The Shape Up Method is a process that helps business owners identify and fix flaws in their products. By using the Shape Up Method, businesses can improve their appeal to customers and increase sales.
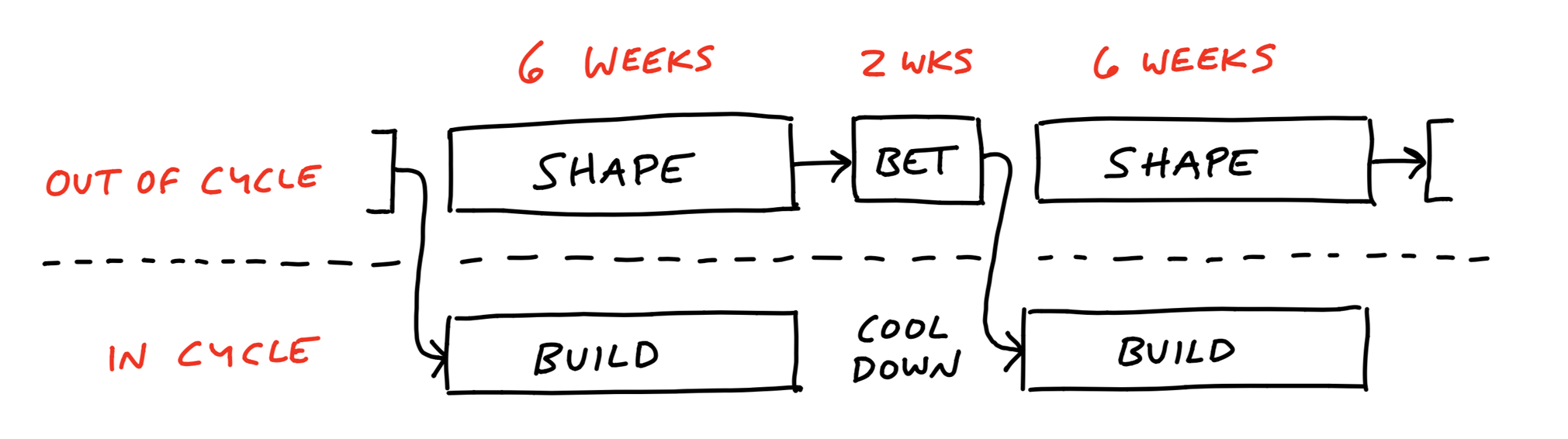
Getting a project done with Shape Up can be divided into three distinct phases:
- Shaping
Those on the senior team, who will not actually execute the project, should be able to define the problem and the solution in the correct manner. As part of the shaping process, the teams need to achieve a balance between being sufficiently explicit, so that the team knows what to do, while not being too prescriptive that they are restricted from figuring out interesting details on their own. Keeping this balance is a delicate process. - Betting
Some senior members of the management team attend such meetings where the shaped projects are discussed and, together, the decision is made whether to approve the projects or reject them. The shaped projects that do not make it through the selection process are not stored in a systematic manner. - Building
The team involved in the shaped project delivers the project with all skill sets present in the team and is fully responsible for the scope in a six-week cycle. Either the project has been completed, or it has been killed, at the end of the six weeks. While extensions are possible, they are exceedingly rare and discouraged.
Shape Up: The Concepts You Need To Know
If you want to be a successful product developer, you need to have a good understanding of basic concepts in shape modeling and optimization. By understanding these concepts, you'll be better equipped to create products that are both efficient and attractive to users.
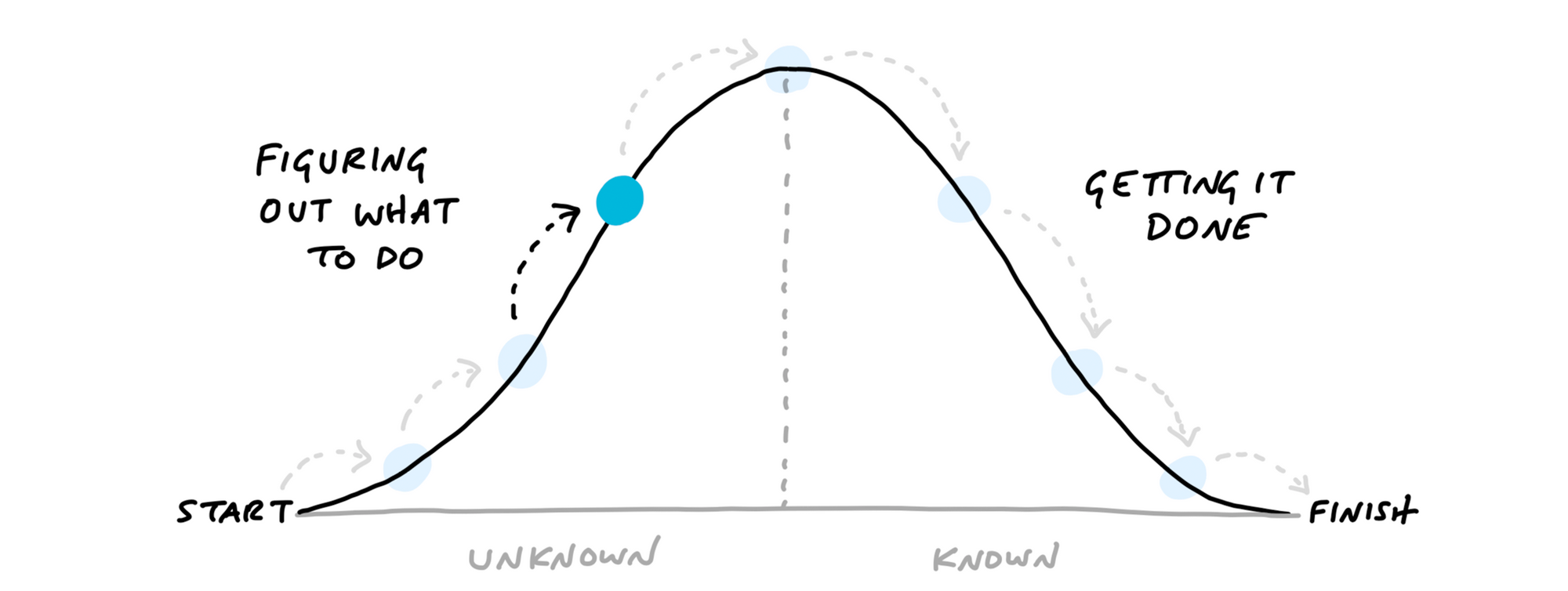
The Shape Up Method was developed by Basecamp to improve productivity and reduce risk in software development. It is centered around following key concepts:
- Six-week work cycle
Work is organized into six-week "sprints" or cycles. This provides a fixed timebox for completing meaningful work rather than open-ended projects. The six-week timeline forces teams to focus and helps identify scope creep. This establishes a realistic time frame for completing a task and provides a firm deadline so that everyone has a clear understanding of how much time they should use each day. - Work shaping
Before each cycle begins, project leaders shape and define the work during a process called "fat marker sessions." This involves outlining key aspects of the project at a high level before the specifics are filled in. Shaping resolves ambiguities upfront rather than leaving teams to flounder. - Team responsibility
Once the shaping is complete, teams are given ownership of the work. They collaborate to define tasks and make adjustments as needed, without micromanagement. This autonomy allows them to do their best work. - Reducing risk
With defined six-week milestones, clear scoping, and team ownership, Shape Up aims to reduce the risk of missed deadlines, undefinable projects, and other pitfalls. - Betting
Management chooses what to work on in each cycle by "betting" on the most important initiatives. This provides focus on priority projects rather than trying to juggle too many. Surveys show most product teams struggle to prioritize effectively.
Shape Up: How To Implement
Product development is an important aspect of any business. It's the process of developing a new product or improving an existing one. There are a lot of different steps that go into product development, and it can be a bit daunting to figure out how to go about it. Here are some tips on how to start product development and get it rolling with the Shape Up method.
The development of a new product is a complex process that can be improved through the use of various tools and techniques. One popular tool for product development is the "Shape Up" technique. This approach helps companies focus on key aspects of their products and develop them in a more efficient manner.
There are several steps that need to be followed when using the Shape Up technique:
- Define the product vision- This step involves developing a clear understanding of what the product should do and what benefits it should provide to customers.
- Create a requirements document- The requirements document defines all of the specific features and functions that the product must have.
- Select a prototype platform- The prototype platform is used to create a working model of the product. It can be anything from a simple sketch to an actual software prototype.
- Develop a product plan- The product plan is used to create a detailed schedule and budget for the project.
- Test and refine the prototype platform- Testing the prototype platform will ensure that all of the functions work as intended.
- Create a finished product- If the prototype platform passes all of its tests, then an actual product can be created.
- Produce the finished product- The final step involves making sure that there aren't any bugs in the product that may cause it to fail.
- Market and sell the product- After a company has created and sold a product, then they can begin to make money.
In order to ensure product success, it is important for companies to have a plan in place for integrating the new product into their business. This plan should include the steps necessary for developing and launching the product as well as how it will be managed after launch. Failing to plan can result in missed opportunities, wasted resources, and delayed time-to-market.
One of the most important steps in product development is market research. This should include studying what products are currently available, what features are being offered, and what customers want. It is also important to understand the company's target market and how the new product will fit into that market. Once the research is complete, a strategy can be developed for bringing the product to market.
The development process itself should be well organized and include specific milestones that need to be met in order to stay on track. Managing the product development process is also important. This includes deciding which products to develop, how they should be marketed, and whether more than one major version of the product will be necessary.
It also involves communicating with the company's partners during each stage of the process. At the end of a product development process, management should have defined the product's features and benefits. The features are those features that will differentiate it from the competition. The benefits are what users will gain from using the new or improved product.
Spend more time planning before you start a project
It's easy to dive in and start working on a project, but spending more time planning can save you a lot of headaches down the road. By taking the time to map out what you want to do and how you're going to do it, you can avoid problems like wasted time and materials, or features that don't work well together.
Project managers use the 6 weeks that Shape Up's teams spend on product development to plan the next 6 weeks by defining a project, defining its boundaries, defining its risks, and determining how it works and where it belongs in the existing process.
It would be ideal to set realistic goals with some abstract elements that would give developers a rough idea of what direction to take, but allow them the freedom to develop it according to their own preferences.
It is important that as soon as the plan is made, it is presented to the team for comments and feedback, which will help identify any missing elements that might have fallen through the cracks.
There are basic benefits to this process, which is to prevent major issues from occurring, such as the scope of the project going out of scope, unforeseen issues slowing down the process, and too many rounds of review/editing in the later stages of the execution.
Ready for more?
We'll dive into the history, background, and answer common questions I get when helping teams set up Shape Up for the first time...
What is the Shape Up Methodology?
The Shape Up Method is a proven system for developing and selling products. It has been successfully used by thousands of product development professionals around the world.
According to the Shape Up method, product development teams shape, bet on, and build meaningful products using specific processes and procedures. The final goal is to ensure teams achieve a great product on time by addressing risks and unknowns at each stage of the product development process and providing teams with language and specific techniques to address these risks and unknowns.
Using Shape Up as a tool, engineers and product managers can improve their own processes and product development results by bridging the gap between product management and the software developers in the field. This is a framework for the development of software products.
In reality, Shape Up is a method that seeks to solve this nagging problem by offering organizations tools that provide a way to bridge the understanding gap, improve project estimations, and prevent projects from tying up too much of their resources.
Shape Up Method: What's its History?
This method was developed by Basecamp based on its own internal product development process. The concept was introduced externally in the book Shape Up: Stop Running in Circles and Ship Work that Matters by Ryan Singer, Manager of Product Strategy at Basecamp.
As expressed by Singer, "one of the core tensions in product management is the tension between the need for detailed implementation details on a day-to-day basis and the need for long-term planning on a strategic level. In the absence of a resolution, this tension leads to missed deadlines, tangled codebases, bad morale, and many other problems.
While Basecamp suffered growing pains on its own, it saw an opportunity to address a new set of challenges that were become increasingly apparent to them on the way (e.g., massive projects without clear boundaries or firm deadlines; long-term strategic considerations overshadowed by day-to-day issues involving product management, and delivering tangible products on time).
With time, Basecamp created what eventually would be called the Shape Up Method, a process for better defining and prioritizing projects before handing them off to the respective teams for development and shipment.
This problem has been plaguing organizations for many years, and Shape Up aims to make it easier for organizations to bridge this understanding gap, estimate their projects more accurately, and make certain projects do not push deadlines to the right.
Is Shape Up an enterprise process framework?
Shape Up is a set of techniques and principles, not a comprehensive process framework like Scaled Agile (SAFe). It does not provide guidance on improving or optimizing development processes, and it's not meant for this. It is a toolbox full of techniques that you can apply as you see fit in your own process.
If you are having problems with your process, Shape Up will not help. It is important to use the right tool for the job, and Shape Up is not the right tool for process improvement.
If you are looking for a process framework there are many process improvement frameworks out there, such as Lean, Six Sigma, and Agile, that are designed to help you improve your process. Shape Up process does not include any of these frameworks, and so it will not help you improve your process.
Is Shaping the same as refinement?
There is a lot of overlap between shaping and refinement, but there are also some key differences. The easier answer is: No, but shaping and refinement are related but distinct processes in the Shape Up method.
Shaping involves initially framing and scoping a project at a high level before handing it off to a team. It focuses on identifying the problem to be solved, articulating the desired outcomes, and outlining guardrails for the team. Shaping is done by product leaders and happens before refinement begins.
Refinement is the iterative process of improving and polishing the specifics of a product once initial shaping has occurred. It involves activities like error fixing, enhancing details, and refining flows. Refinement happens during development by the hands-on team.
While shaping and refinement have some overlap, they serve different purposes. Shaping provides the initial structure and direction. Refinement then builds on that foundation through incremental enhancements during execution. Both are critical to developing a high-quality product.
The key difference is that shaping is broad framing done before work begins, while refinement is detailed improvements done during development. Shaping sets the stage, refinement brings it to life. Both are essential phases in the Shape Up workflow.
In Shape Up, what does it mean to bet?
Shape Up is a term used in business for the act of betting or choosing which projects get a team to work on for a cycle of 6 weeks. It's a commitment by leadership to fully support that project for the duration.
A group of important stakeholders come together and decide which project they feel is most important and should be given the most attention.
This betting process isn't without risks. Leaders have imperfect information and must make judgment calls. Some bets may not pay off. However, when done right, betting aligns the organization and focuses efforts on the projects that matter most. It gives teams clarity on what to build and why it's important.
The betting phase is crucial for strategic alignment. By betting on the right projects, leaders point their teams toward delivering meaningful outcomes that advance the company's goals.
This usually means that the team working on this project will have to be dedicated to it full time for the next six weeks. It can be a risky move, but if it pays off, the company can see significant improvements.
Recommendation
I recommend Shape Up consistently to early-stage startups, companies under 250 people, and small teams within an Enterprise with the autonomy to set their own work methods – it empowers autonomous teams and cuts the BS out of most Agile Scrum overhead when companies are small.
The Shape Up Method provides product managers and engineers with a revolutionary new approach to the product development process.
Shaping and better were originally intended for senior startup founders, but can easily be mapped to product managers.
By embracing key concepts like six-week cycles, work shaping, betting, and six-week sprints, teams can transform the way they build successful, meaningful products.
The Shape Up Method replaces undefined scopes and unspecified solutions with a process focused on rapid iteration and meaningful problem solving.
With the strategies outlined in this guide, product teams can master the Shape Up Method for product engineering, prevent scope creep, and deliver innovative solutions iteratively.
By adopting the Shape Up Method, any team can level up their engineering team practices and build successful, meaningful products iteratively and efficiently.
Your team can start shaping better products today.
FAQ for Shape Up
What is the purpose of the six week cycle in Shape Up?
The six week cycle is a core component of the Shape Up methodology developed by Basecamp. This provides a fixed timebox for teams to complete meaningful work rather than open-ended projects.
The six week timeline forces product teams to focus their efforts and helps identify scope creep.
The 6-week cycle also establishes a realistic timeframe for software development teams to ship a meaningful product increment.
At the end of each six week cycle, teams demo their work and then the process repeats.
Why are six weeks important?
Six weeks is the ideal length of time for product teams using Shape Up. It is short enough to maintain focus but long enough to build something significant. Six weeks also sync up well with common software development sprints. Having set six week cycles also makes planning easier since you know how much can get done in each chunk of time. Overall, the recurring six week timeline is key for shipping incremental progress.
What happens at the end of a six week cycle?
At the end of each six week cycle, the product team demos their completed work for the senior team members. This demo session is crucial for getting feedback and aligning on what should happen in the next cycle.
After the demo, the development team takes a break from actively building as the product enters a "cool down" phase.
This lets them recharge before betting on what to build in the next cycle begins.
How does the betting table work?
The betting table is where senior team members come together to decide what the product team will work on in the next cycle. They pitch ideas and make the case for which projects are most important for the next six weeks.
After discussion and debate, they place bets on the ideas they want to prioritize.
This aligns the organization around the most critical initiatives. The product team then builds what was "bet on" during the next six week cycle.
What are the key benefits of shaping projects?
Shaping projects before a six week cycle starts is crucial. In a shaping session, product leaders define and scope the work to be done at a high level.
This upfront shaping resolves ambiguities so teams don't lose time clarifying details later. It also prevents teams from going down rabbit holes.
Overall, shaping sets up product teams for a successful six week period.
Why is the final phase of each cycle a cool down?
The cool down period after the six week cycle ends gives the product team time to recover and wrap up loose ends.
It is also when the senior team can pitch ideas for the next cycle. Cool down prevents burnout by giving development teams a break between intense sprints.
This lets important ideas percolate before betting begins again. This ensures teams stay fresh.